
| Research Abstracts Home | CSAIL Digital Archive | Research Activities | CSAIL Home |
![]()

|
Research
Abstracts - 2007
|
Blood Glucose Monitoring in Critical CareYing Zhang & Peter SzolovitsMotivationThe stress from critical illnesses induces both insulin resistance and unsuppressed gluconeogenesis, necessitating blood glucose monitoring in over 90% of the patients in the Intensive Care Unit (ICU). For decades, a moderate level of this stress-induced hyperglycemia was considered a beneficial compensatory mechanism, and the conventional insulin therapy for adults worked to maintain blood glucose level in the 180-215 mg/dL range. Recently, however, a number of randomized, controlled clinical trials have demonstrated that strict control of the normoglycemic range of 80-110 mg/dL reduces both in-hospital mortality and morbidity for adult patients who stayed in the surgical ICU for more than five days and those who stayed in the medical ICU for more than three days. [1, 2] Though the past few years have seen many new efforts to control blood glucose levels, protocols to implement strict insulin therapy, like those for conventional therapy, often generate rapid, seesaw-like changes in blood glucose levels. They maintain patients within the target range for only a less-than-desirable fraction of the time. [3] Achieving tighter control is made difficult because (a) existing protocols rely on pre-selected trigger glucose levels, but todays workload on ICU staff enables neither more frequent blood glucose measurements nor real-time tracking of protocol compliance and effectiveness; (b) the trigger glucose level is usually set at or above the upper bound of the normoglycemic range because intensive insulin therapy puts patients at a higher risk for acute hypoglycemia, which can cause permanent brain damage or even death in the presence of hypoxia or severe comorbidities; [4] (c) the growing emphasis on providing adequate nutritional support during critical care imposes further constraints. MethodOur first step to help improve glycemic control in critical care is to utilize the rich clinical data repository of a comprehensive and integrated ICU database called MIMIC II (Multiparameter Intelligent Monitoring in Intensive Care). [5] This database consists of recordings from the bedside monitoring devices and the clinical information systems at the CCU (Coronary Care Unit), Medical ICU, and Surgical ICU of a tertiary teaching hospital. It includes continuous high resolution physiologic waveforms such as ECG, vital signs, monitoring alarms, therapeutic intervention profiles, laboratory results, fluid balance, continuous IV medications, physician order entries, nursing progress notes, discharge summaries, and patient outcomes. After observing how blood glucose levels are monitored in patients at a medical ICU, we examine blood glucose measurements and IV insulin administration recordings in the MIMIC II database. For patients on IV insulin, we focus on the changes in blood glucose levels over time in response to IV insulin therapy. ResultsBlood glucose levels are routinely measured during intensive care. Of the 17242 patients for whom we have laboratory values in the MIMIC II database, over 93% had blood glucose measurements. The usual method of measurement is by a fingerstick glucose meter. Once a decision to initiate antihyperglycemic therapy is made, caregivers follow a hospital-specific protocol, along with more frequent blood glucose measurements at pre-defined time intervals: every 1 hour when the patient is on IV insulin, every 4 hours when the patient is dosed according to an insulin sliding scale. Preliminary findings indicate that records of blood glucose measurements are not consistently available at the pre-defined time intervals, but if they are available and the patient was on IV insulin, most of them fall within the pre-defined intervals by a factor of 2 (e.g. ranging from 30 minutes to 2 hours). During IV insulin therapy, all patients except one received close blood glucose monitoring. The exception is a case where although IV insulin was initiated based on patient history and presentation, the patient expired shortly after admission and before any blood glucose measurements could be performed. For 3117 patients, we examine how blood glucose level had changed over time and tracked IV insulin administration in correlation with the blood glucose data. Figure 1 shows the typical course of a patients glycemic state over time in response to IV insulin therapy. 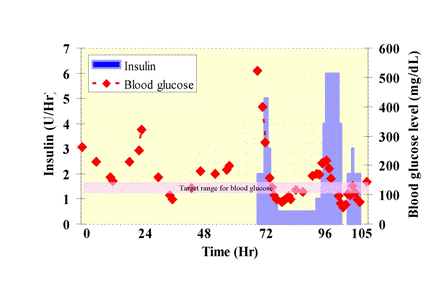
Figure 1. Glycemic state over time. The infusion protocol used at the teaching hospital has a target range (marked by the pink band) of 101 -150 mg/dL for blood glucose. DiscussionsBlood glucose monitoring is an integral part of critical care, as evidenced by the high percentage of patients being monitored in our study. While the means of measurement is quick and accessible, the frequency at which blood glucose must be measured is high. The observed course and variance in blood glucose level includes contributions from physiological factors such as the therapy regiment and non-physiological factors such as the frequency of measurements. Figure 1 shows the two key features of glycemic state over time: 1) the proportion of time in which blood glucose is within the normoglycemic range is small; 2) IV insulin induces seesaw-like changes in blood glucose levels. These observations verify what we have found in the literature. In addition, changes in the blood glucose level lag the IV insulin administration on the order of a couple of hours. This observed delay may be an artifact generated by the frequency of blood glucose measurements. The contributions from the onset time for the insulin and the diffusion time of insulin from the circulation to the extra cellular space are also worth further investigation. The next step of this research is to model glucose uptake and insulin resistance based the underlying pathophysiology and use a combination of learning and inference techniques to track patients glycemic states. After incorporating physician order entries into constructing glycemic profiles, we will extend our methods to patients who are being dosed according to an insulin sliding scale. Funding Source(s)This work is supported by NLM Medical Informatics Training Grant. References:[1] Greet Van den Berghe, Pieter Wouters, Frank Weekers, et al. Intensive insulin therapy in critically ill patients. In N Engl J Med, 345(19), pp. 1359-1367, 2001. [2] Greet Van den Berghe, Alexander Wilmer, Greet Hermans, et al. Intensive insulin therapy in the medical ICU. In N Engl J Med, 354(5), pp. 449-461, 2006. [3] Justine M. Carr, Frank W. Selke, Michelle Fey, et al. Implementing tight glucose control after coronary artery bypass surgery. In Ann Thorac Surg, 80, pp. 902-909, 2005. [4] Anuj Bhatia, Brit Cadman, and Iain Mackenzies. Hypoglycemia and cardiac arrest in a critically ill patient on strict glycemic control. In Anesth Analg, 102, pp. 549-551, 2006. [5] Brian A. Janz, Mohammed Saeed, Gari D. Clifford, Roger G. Mark, et al. MIMIC II: A massive temporal database to support research in integrating data, models, and reasoning in critical care. In Proceedings of AMIA 2005 Fall Symposium, Washington DC, USA, October 2005. |
||||
|